
UV/H2O2 and UV/PDS Treatment of Trimethoprim and Sulfamethoxazole in Synthetic Human Urine: Transformation Products and Toxicity | Environmental Science & Technology
![PDF) Synthesis, structure and thermal decomposition of [Ni(NH3)6][VO(O2)2( NH3)]2 | Jana Chrappová - Academia.edu PDF) Synthesis, structure and thermal decomposition of [Ni(NH3)6][VO(O2)2( NH3)]2 | Jana Chrappová - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/42328747/mini_magick20190217-5564-bhy5f6.png?1550465508)
PDF) Synthesis, structure and thermal decomposition of [Ni(NH3)6][VO(O2)2( NH3)]2 | Jana Chrappová - Academia.edu
2 and [Fe(NH3)6SO4]CN are the ionization isomers of the [Ni(en)2(NO2)2]Cl2 and [Fe(NH3)6CN]SO4 respectively.If true enter 1, else enter 0. Ni(en)2Cl2](NO2)2 and [Fe(NH3)6SO4]CN are the ionization isomers of the [Ni(en)2(NO2)2]Cl2 and [Fe(NH3)6CN]SO4 respectively.If true enter 1, else enter 0.](https://haygot.s3.amazonaws.com/questions/279979_306961_ans.png)
Ni(en)2Cl2](NO2)2 and [Fe(NH3)6SO4]CN are the ionization isomers of the [Ni(en)2(NO2)2]Cl2 and [Fe(NH3)6CN]SO4 respectively.If true enter 1, else enter 0.
![Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex:(i) K[Cr(H2O)2 (C2O4)2].3H2O ( Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex:(i) K[Cr(H2O)2 (C2O4)2].3H2O (](https://haygot.s3.amazonaws.com/questions/1970729_1266809_ans_fecfd8ebb95e482282431be5eeccd43e.jpg)
Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex:(i) K[Cr(H2O)2 (C2O4)2].3H2O (
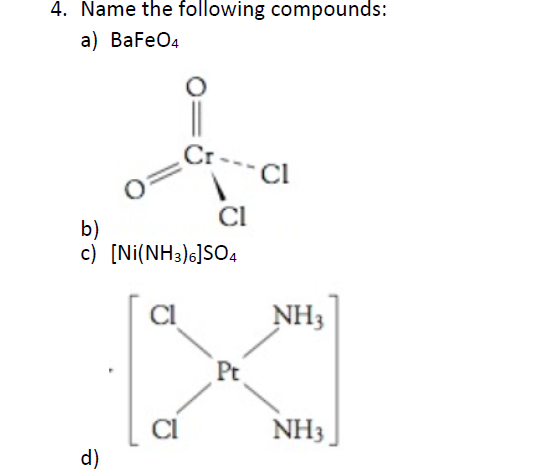
![Using IUPAC norms write the systematic names of the following:(i) [Co(NH3)6]Cl3 (ii) [Pt(NH3)2Cl(NH2CH3)]Cl (iii) [Ti(H2O)6]^3 + (iv) [Co(NH3)4Cl(NO2]Cl (v) [Mn(H2O)6]^2 + (vi) [NiCl4]^2 - (vii) [Ni(NH3)6]Cl2 (viii) [Co(en)3]^3 + (ix) [Ni(CO)4] Using IUPAC norms write the systematic names of the following:(i) [Co(NH3)6]Cl3 (ii) [Pt(NH3)2Cl(NH2CH3)]Cl (iii) [Ti(H2O)6]^3 + (iv) [Co(NH3)4Cl(NO2]Cl (v) [Mn(H2O)6]^2 + (vi) [NiCl4]^2 - (vii) [Ni(NH3)6]Cl2 (viii) [Co(en)3]^3 + (ix) [Ni(CO)4]](https://haygot.s3.amazonaws.com/questions/1932453_1846912_ans_48f35b4ba31346d190946b3438b03b2c.jpg)
![PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2 PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2](https://www.researchgate.net/profile/M-Drabik/publication/256461217/figure/tbl1/AS:667594596024340@1536178363556/Characteristic-bands-in-infrared-spectrum-of-NiNH3-6-VOO-2-2-NH-3-2_Q320.jpg)

![PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2 PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2](https://www.researchgate.net/profile/M-Drabik/publication/256461217/figure/fig1/AS:525770654011392@1502364900131/ray-powder-diffraction-patterns-of-Ni-2-V-2-O-7-15-V-2-O-5-and-intermediate-products_Q320.jpg)

2 с K4[Fe(CN)6] - YouTube Химическая реакция [Ni(NH3)6](OH)2 с K4[Fe(CN)6] - YouTube](https://i.ytimg.com/vi/wTdaG_zBn8A/maxresdefault.jpg)


![PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2 PDF) Synthesis, structure and thermal decomposition of [Ni(NH3) 6][VO(O2)2( NH3)]2](https://i1.rgstatic.net/publication/256461217_Synthesis_structure_and_thermal_decomposition_of_NiNH3_6VOO22NH32/links/598c15b1458515c333a5e1ff/largepreview.png)





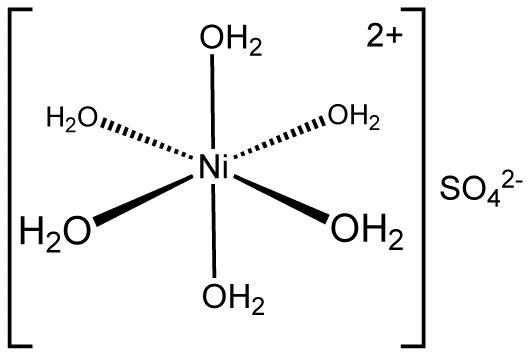
![inorganic chemistry - Confused on how to draw cis-[Ni(H2O)4(NH3)2]Cl2 - Chemistry Stack Exchange inorganic chemistry - Confused on how to draw cis-[Ni(H2O)4(NH3)2]Cl2 - Chemistry Stack Exchange](https://i.stack.imgur.com/zOmAk.png)
![the starting material is [Ni(NH3)6]SO4, and the final | Chegg.com the starting material is [Ni(NH3)6]SO4, and the final | Chegg.com](https://media.cheggcdn.com/media/d62/d6298c59-34a8-48f2-b833-3875f63e7462/phpgtLVGs.png)